 
Let's talk about a few more of these rules. We can summarize this as follows, ionic compounds with ions of low charge are often soluble in water. If we relate it to Coulomb's Law, the cation/anion attractive forces are relatively small when the charge values are small, and here we've got charge values of either plus 1 or minus 1, depending on which rule we're looking at. It comes down to Coulomb's Law once again. All of those compounds are soluble in water. So you could have, for example, aluminum acetate, or lead two nitrate, or copper one perchlorate. If you have a whole bunch of nonmetals grouped together, and that group of atoms has a negative 1 charge, and here's some examples, acetate, perchlorate, chlorate, and nitrate, then the ionic compounds of those species are soluble. What does that mean? Well, here's some examples. This rule is summarized as, compounds of many atomed, low charged polyatomic ions are soluble. Rule number two is now looking at it from the perspective of the anion, whereas rule number one listed a bunch of cations, rule number two lists a bunch of anions. So, most of the time, ammonium compounds, sodium compounds, lithium compounds, potassium compounds, anything covered by rule number one, have high solubility in water. Exceptions to rule number one are fairly rare. All of those have a plus 1 charge as their most frequent oxidation state. Now, if you don't have a periodic table handy, you might not remember that the 1A metal cations include lithium, sodium, potassium, cesium, and rubidium. The first rule is that compounds of ammonium ion, or the 1A metal cations, are soluble. I'm going to list them here and then we'll do some examples. These rules are also summarized on the reference page of the course homepage. You can find various solubility rules listed in general chemistry textbooks or on webpages throughout the internet. So let's keep Coulomb's Law in mind as we talk about the solubility rules. One of the ways that can happen with ionic compounds is to have an ion that has a relatively large atomic radius, or to have a polyatomic ion which has many atoms together with, for example, a minus 1 charge, spread out over a large distance. So if you have a large distance between the particles, then the force of attraction is small. Distance, on the other hand, is in the denominator. If we increase the magnitude of the charges, for example, if we go from plus 1 for an alkaline metal to plus 2 for an alkaline earth metal or plus 3 for aluminum, for example, then the force of attraction becomes stronger, because the charges are on the numerator. If the charges q1 and q2 are small in magnitude, in other words, if their absolute value is one, then we expect the force of attraction to also be relatively small. 
Carbon disulfide has a dielectric constant of only 2.6, which is a lot lower than 80. 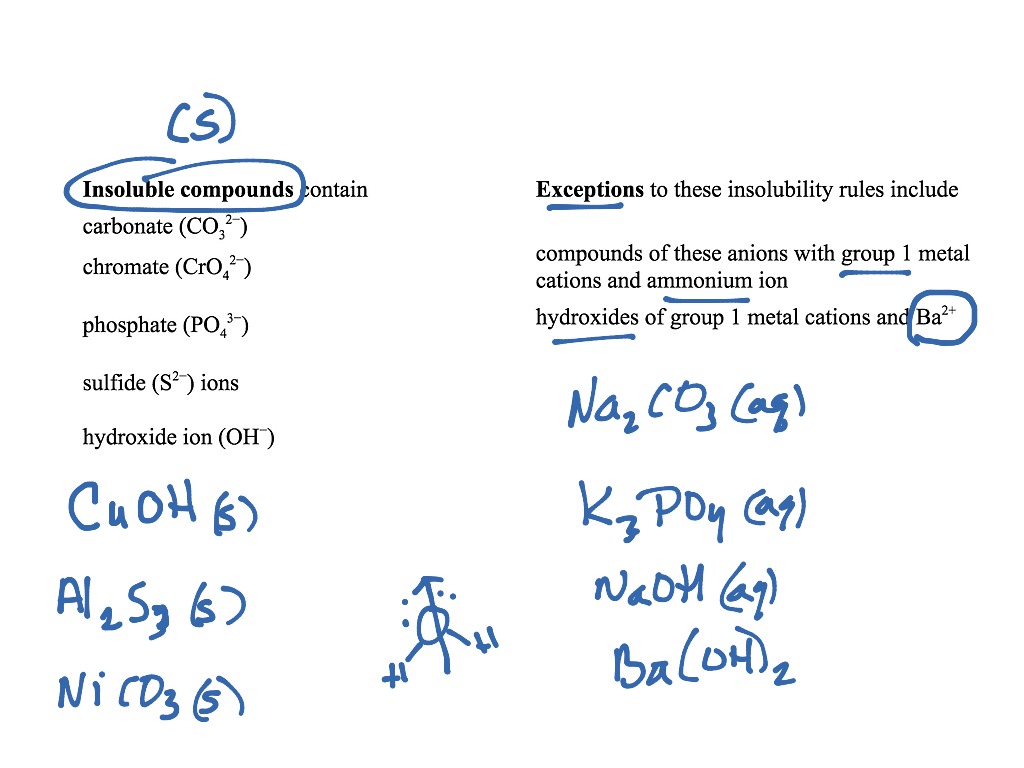
For example, air has a dielectric constant of about one, and even some other liquid solvents, like some hexane or some carbon disulfide, have relatively low dielectric constant. 
That means that the force of attraction between ions and water is low relative to what that force of attraction would be in a lower dielectric medium. Because the dielectric constant is in the denominator, and the dielectric constant of water at room temperature is about 80, we'll be dividing whatever is in the numerator by a factor of 80, times the distance squared. One of the first things I'd like to point out when we're discussing Coulomb's Law in the context of solutions, is that for aqueous solutions, we will have water as the solvent, and the dielectric constant of water is relatively high. Remember, Coulomb's Law dealt with the attraction or repulsion between charged particles. But without doing the experiments ourselves, without looking at all the different ionic compounds that are available, and trying to see how well they dissolve in water, how can we predict whether or not a particular ionic compound will dissolve in water? Well, predicting something like this takes us all the way back to one of the first things we've discussed in this course, and that is, Coulomb's Law. We determined that if the concentration of ions can be above 0.1 molar, then that species was defined as being soluble in that solvent. You can do this for other solvents as well, but in the next few lectures, we'll be considering aqueous solutions. Solubility for ionic compounds was related to the amount of solid that would dissolve in the water. First, I want you to recall the definitions of solubility. In this lecture, I'll be discussing solubility rules for ionic compounds dissolving in water. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
